Categories Products Center
Contact us
ZHEJIANG JHENTEN MACHINERY CO.,LTD.
Tel:+86-577-86867777
Fax:+86-577-86810195
E-mail:info@jhenten.com
Overview
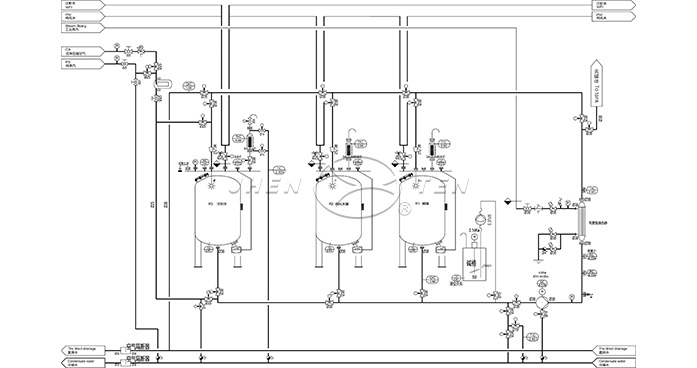
Key Process Control Points
Precision Cleaning Control◆Multi-stage programs:
·Pre-rinse → alkaline wash → acid wash → final rinse
·Customizable sequence switching
◆Critical parameter monitoring:
·Temperature
·Turbulent flow
·Concentration
Sterilization Efficacy Assurance
◆Pure steam sterilization (SIP)
·Temperature-pressure-time validation
·Biological indicator testing
·Overheated water sterilization

Cleaning Media Management
◆Smart dosing
·Closed-loop control of concentrations
◆Recycling
·Reuse filtered solutions (pharma) or neutralize discharge (food)
Data Integrity & Traceability
◆21 CFR Part 11-compliant electronic records
·Automated cleaning/sterilization reports.
·Generate audit-ready batch reports with a single click.
◆Audit trails
·Logs for parameter adjustments, operator privilege changes, and system alarms.